For QMS managers tired of chasing impact across silos
AI-Powered eQMS
for Medical Device Companies
Built for
ISO 13485, FDA QMSR, EU MDR, IVDR, and SaMD workflows.
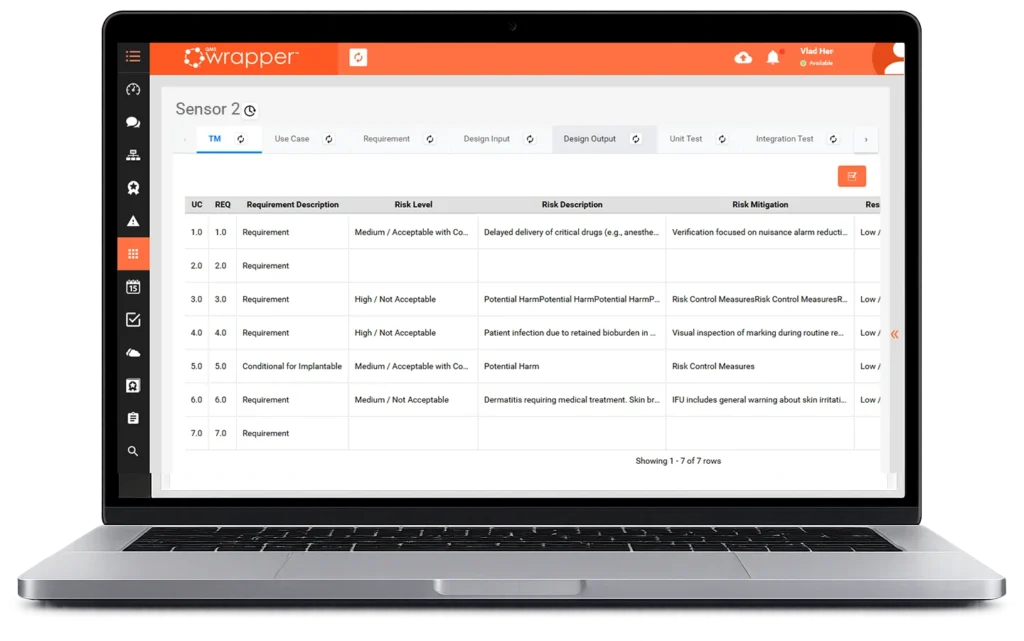
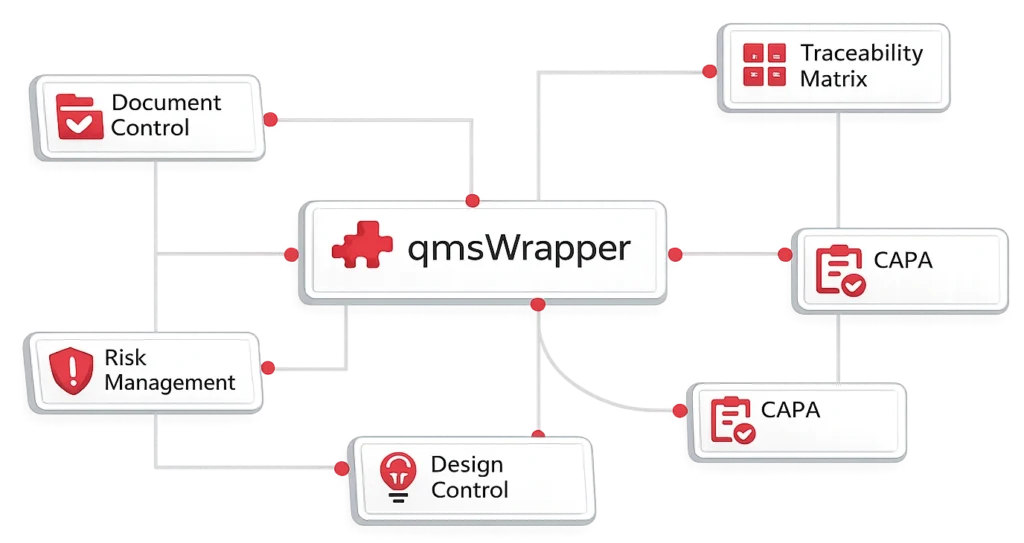
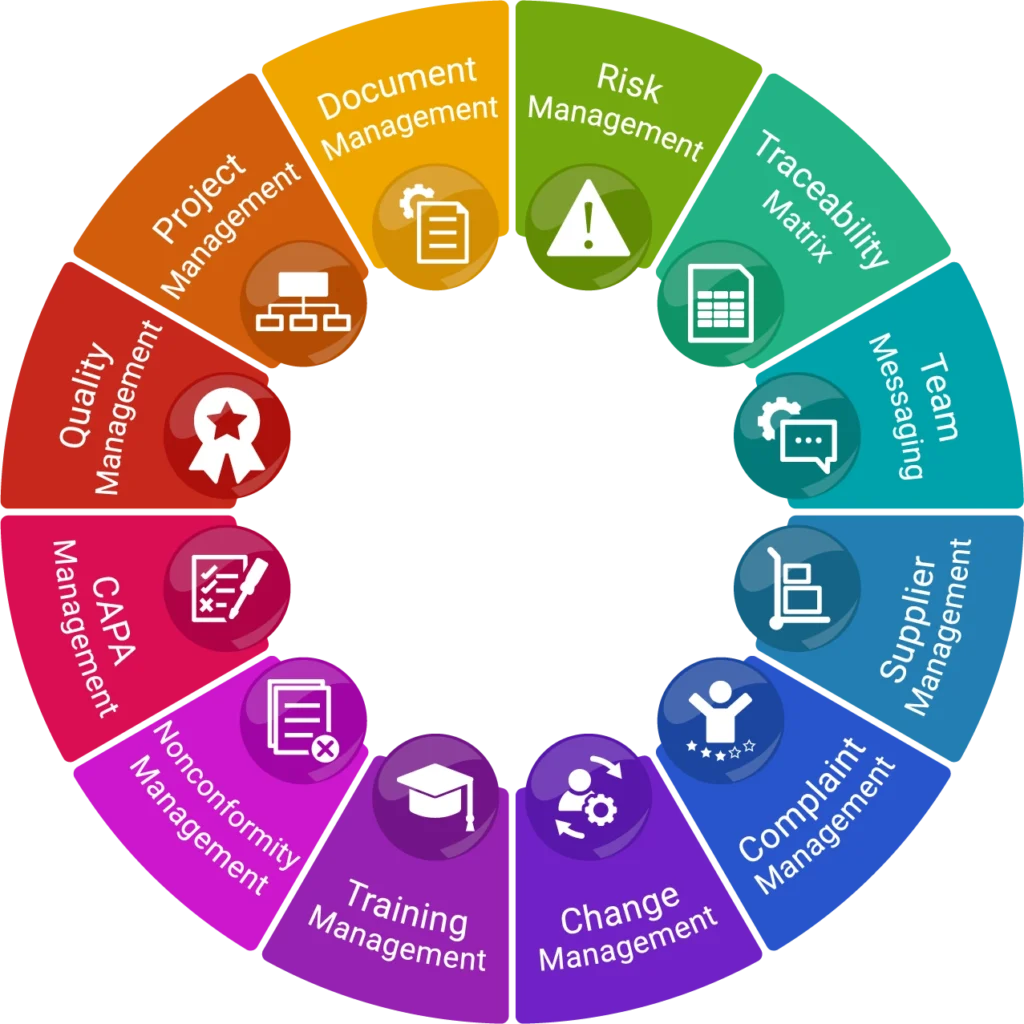
qmsWrapper helps MedTech teams understand how quality events, changes, CAPAs, risks, requirements, and Technical File records are connected, before compliance gaps appear.
AI-powered workflows connect impact analysis, traceability, and audit-ready documentation across the entire product lifecycle.
“For quality managers preparing for FDA or MDR inspections:
qmsWrapper automates traceability from quality event to technical file update,
cutting manual impact analysis by over 70% while ensuring continuous audit-readiness.”