
qmsWrapper Triumphs with G2 Badges: Empowering Europe's QMS Leaders
Software for quality management systems (QMS), qmsWrapper, is pleased to announce that it has recently earned two prestigious badges from G2 for the Summer 2023 season. Because of its dedication to client satisfaction and distinctive collection of features, qmsWrapper stands out in the market.

qmsWrapper's Badge: High Performer Europe - Summer 2023
One of the honors qmsWrapper received this summer is High Performer Europe - Summer 2023. This acknowledgment shows that qmsWrapper excels beyond its competitors in terms of high customer satisfaction ratings and a steadfast dedication to quality management. Our software has repeatedly shown its capacity to improve cooperation, streamline operations, and provide great traceability features, making it a useful resource for enterprises in Europe and beyond.
Benefits of qmsWrapper:
Enhanced Efficiency and Audit Reduction:
Andy Fisher, Senior Developer at Breathe Suite, attests to the significant reduction in on-site audit time achieved through qmsWrapper. By providing auditors with access to review documents within the software, qmsWrapper has empowered organizations to streamline their audit processes, resulting in more efficient operations.
Expertise Tailored for Medical Devices:
Vicky Chen from Simple HealthKit appreciates qmsWrapper's unique advantage of being developed by medical device professionals. This key differentiator ensures that the software is designed with deep industry knowledge and understanding, catering specifically to the needs of medical device companies.
Easy Data Management and Traceability:
Joshua Park, CEO of Globe Biomedical, highlights qmsWrapper's ability to manage large amounts of data while maintaining traceability. The software's intuitive interface enables seamless collaboration and fosters teamwork, facilitating effective quality management within organizations.
Comprehensive Regulatory Compliance:
Kyle Mohney, VPO at Liberty Vision, describes qmsWrapper as a game-changer, offering everything needed for success in regulatory affairs. The software provides comprehensive support, educational materials, and affordable pricing options, making it accessible to both established companies and new medical device ventures.
Pre-built Standard Requirements:
Maaike de Jong, QM at Scinvivo, appreciates that qmsWrapper incorporates standard requirements into the system, eliminating the need for manual configuration. This feature allows businesses to quickly adapt to industry standards while saving valuable time and resources.
Guided Design Control and Risk Management:
Sue Bithel, CTO of Calon Cardio, highlights qmsWrapper's supportive approach to design control and risk management processes. The software provides step-by-step guidance, offering invaluable assistance to companies with limited experience in these areas. While hiring a full-time quality manager is still recommended, qmsWrapper serves as a reliable resource for organizations seeking to navigate these processes effectively.
High Performer Europe - Summer 2023 is one of the awards that qmsWrapper is happy to have received for this summer. This acknowledgement shows that qmsWrapper excels beyond its competitors in terms of high customer satisfaction ratings and a steadfast dedication to quality management. Our software has repeatedly shown its capacity to improve cooperation, streamline operations, and provide great traceability features, making it a useful resource for enterprises in Europe and beyond.
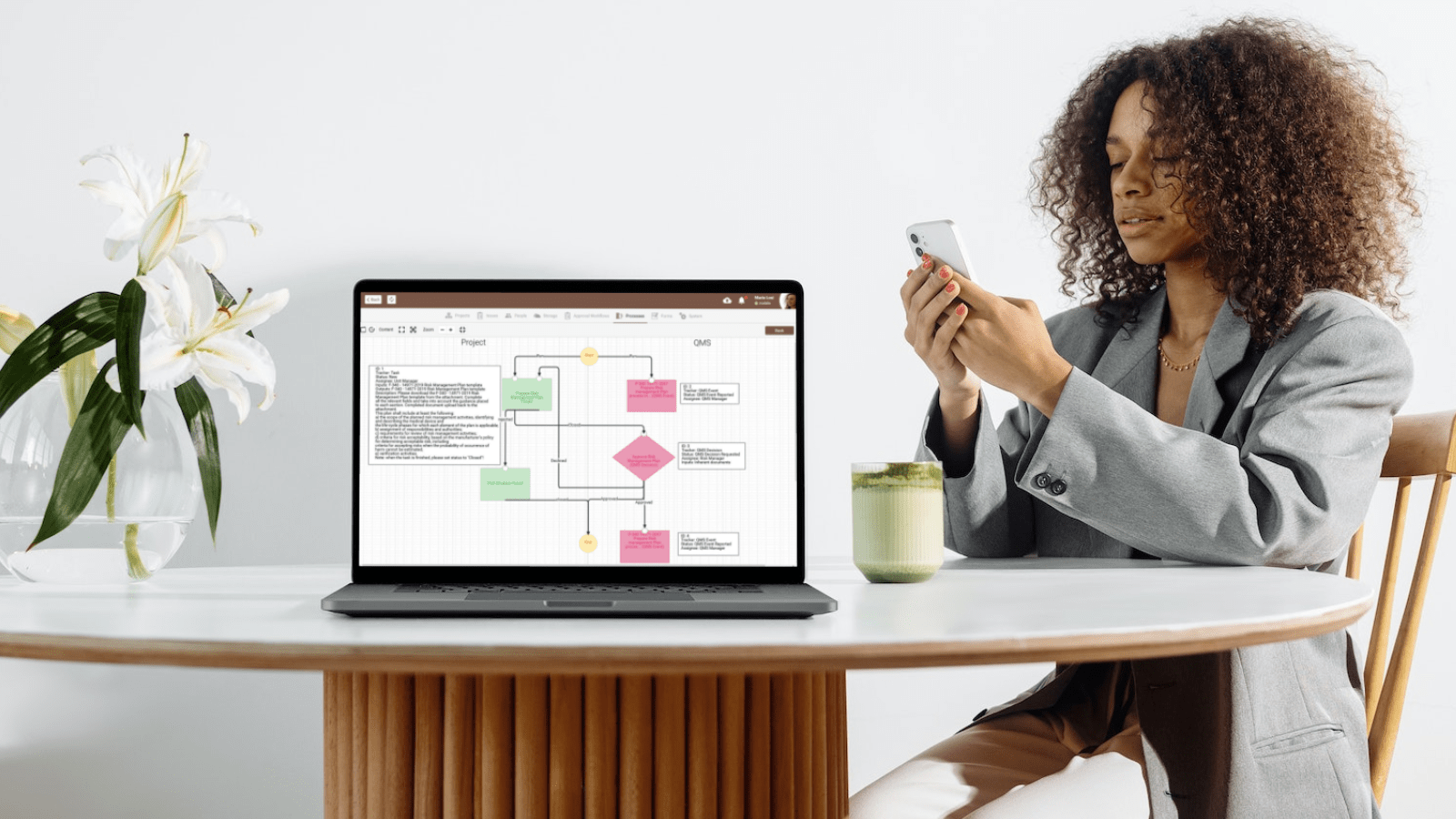
We cordially invite everyone who hasn't yet benefited from qmsWrapper to take the next step in streamlining their quality management procedures. To see firsthand how qmsWrapper can improve your company's approach to quality management, schedule a demo now or contact us at contact@qmswrapper.com. Don't pass up the chance to use qmsWrapper's cutting-edge software to improve communication, streamline processes, and achieve regulatory excellence. Get in touch with us right away to start your journey to unrivaled quality management achievement.
A New Vision for Risk-Driven QMS with qmsWrapper
qmsWrapper Recognized as a Rising Star in the Spring 2025 Quality Management Software Customer Success Report
Enhancing Visibility and Accountability in QMS with qmsWrapper’s New Log Functionality